The issue price is the price at which the bond issuer originally sells the bonds.The maturity date is the date on which the bond will mature and the bond issuer will pay the bondholder the face value of the bond.Payments can be made in any interval, but the standard is semiannual payments. Coupon dates are the dates on which the bond issuer will make interest payments. If the attractive forces between two or more atoms are strong enough to make them into an enduring unit with its own observable properties, we call the result a 'molecule' and refer to the force as a 'chemical bond'.For example, a 5% coupon rate means that bondholders will receive 5% x $1,000 face value = $50 every year. Section III contains a critical survey concerning the limited possibility of defining bond dipole moments in gen- eral and the practicality of such a definition. 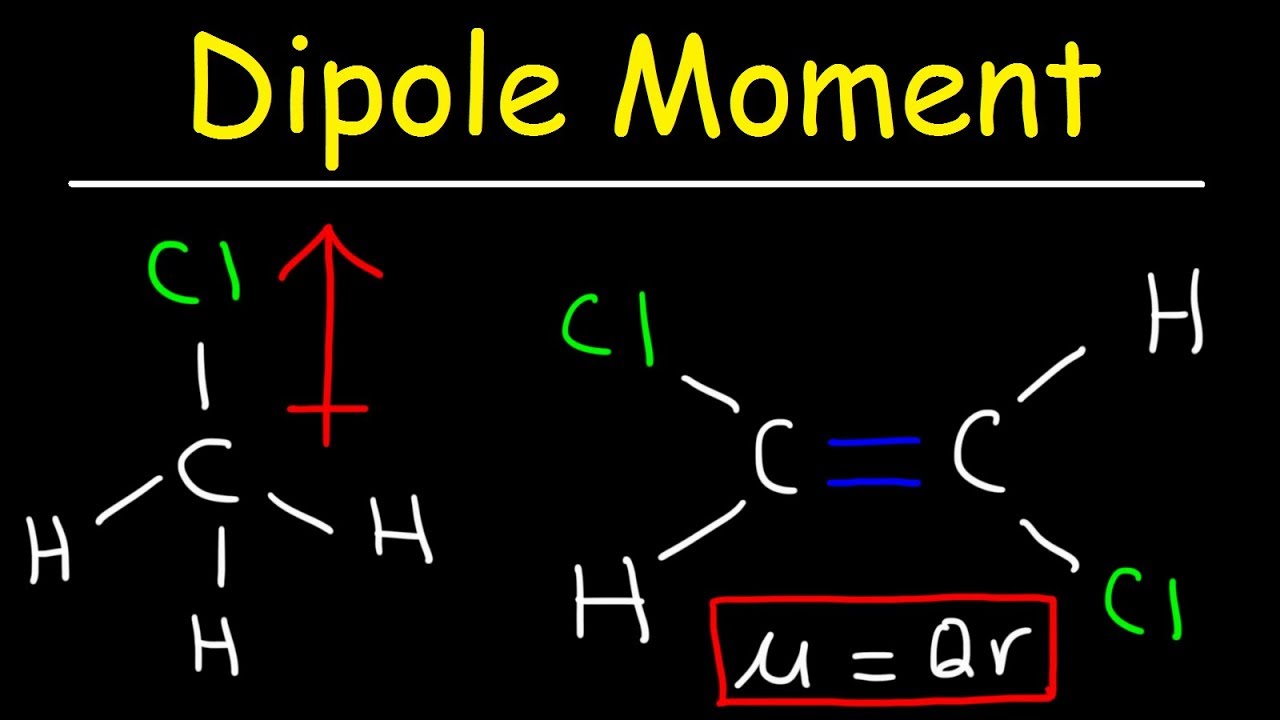
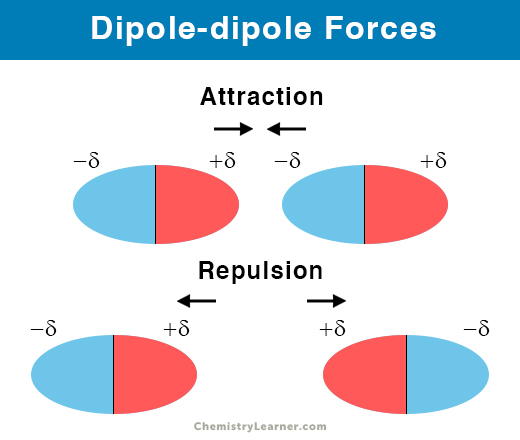
The coupon rate is the rate of interest the bond issuer will pay on the face value of the bond, expressed as a percentage.For example, the dipole moment of halogenhydrogen bonds increases. When the bond matures, both investors will receive the $1,000 face value of the bond. Thus, the electric dipole moment characterizes the degree of ionicity of a chemical bond. For example, say an investor purchases a bond at a premium of $1,090, and another investor buys the same bond later when it is trading at a discount for $980. Table A2 shows the electronegativity of some of the common elements. The vector points from positive to negative, on both the molecular (net) dipole moment and the individual bond dipoles. Physicist tend to use the opposite orientation. The existence of a dipole moment is the difference between polar and nonpolar bonds. The convention in chemistry is that the arrow representing the dipole moment goes from positive to negative. Face value (par value) is the money amount the bond will be worth at maturity it is also the reference amount the bond issuer uses when calculating interest payments. In chemistry, dipole moments are applied to the distribution of electrons between two bonded atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
